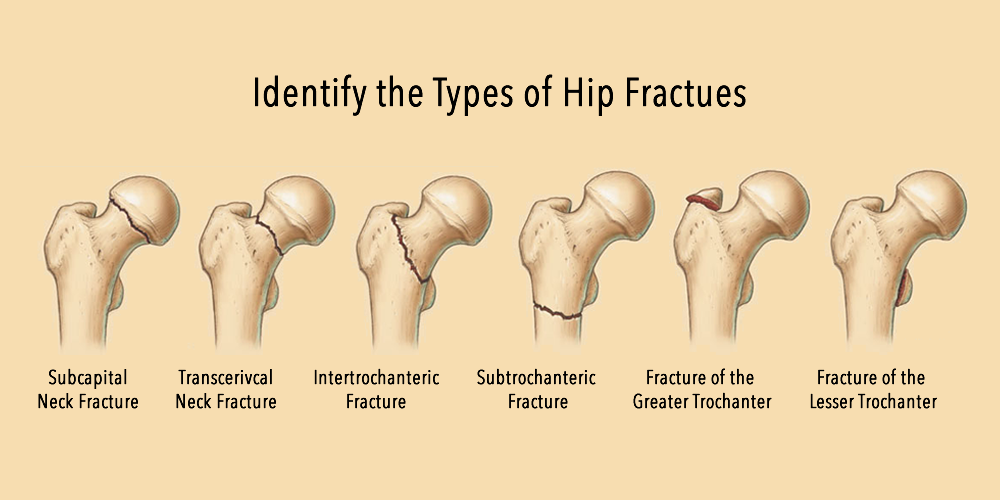
Rotator Cuff
Orthopedic/Rheumatology
BioSolutions Open Protocol Summaries
INDICATION
AGE CRITERIA
DESCRIPTION
40-75, inclusive
RC-002: A Prospective, Double-Blinded, Multi-Center Pivotal Trial of Autologous Adult Adipose-Derived Regenerative Cell Injection into Chronic Partial-Thickness Rotator Cuff Tears
OA Knee
> 40 yrs
A Phase 3, Randomized, Double-blind, Placebo- and Active-controlled Study to Evaluate the Efficacy and Safety of TLC599 in Patients with Osteoarthritis of the Knee
OA Knee
40-80, inclusive
Samumed, LLC/ “A study to support the radiographic eligibility screening process of samumed studies in symptomatic knee osteoarthritis subjects”
Rheumatoid Arthritis
18 years or greater
A Randomized, Controlled Pragmatic Phase 3b/4 Study of Baricitinib in Patients with Rheumatoid Arthritis
OA Knee
40-80, inclusive
A Phase 3, 28-Week, Multicenter, Randomized, Double-Blind, Placebo-Controlled Study to Evaluate the Efficacy and Safety of a Single Injection of SM04690 Injected in the Target Knee Joint of Moderately to Severely Symptomatic Osteoarthritis Subjects
OA Knee
Pts who participated in IA UBX0101 study
Long-term follow-up study of patients with moderate to severe, painful osteoarthritis of the knee who participated in a randomized, placebo-controlled study of UBX0101
BioSolutions Start-Up (Upcoming) Protocol Summaries
INDICATION
AGE CRITERIA
DESCRIPTION
OA Knee
≥ 40 yrs. old
A Randomized, Double-Blind, Placebo-Controlled, Multi-Center, Phase 3 Study to Determine the Efficacy and Effectiveness of TG-C in Subjects with Kellgren and Lawrence Grade 2 or 3 Osteoarthritis of the Knee
Cartilage repair
18-60
A prospective, randomized, active treatment-controlled, evaluator-blinded (radiologist reviewer and physician evaluator) multicenter study to establish the superiority of a hyaluronan-based scaffold (Hyalofast®) with autologous bone marrow aspirate concentrate (BMAC) in the treatment of articular knee cartilage defect lesions in comparison to control (microfracture treatment)
OA Knee
18 years of greater
A Multi-Center, Active-Controlled, Open-Label, Phase 2 Trial to Compare the Efficacy adn Safety of CartiLife, and Microfracture for Patients with Articular Cartilage Defects in the Knee